 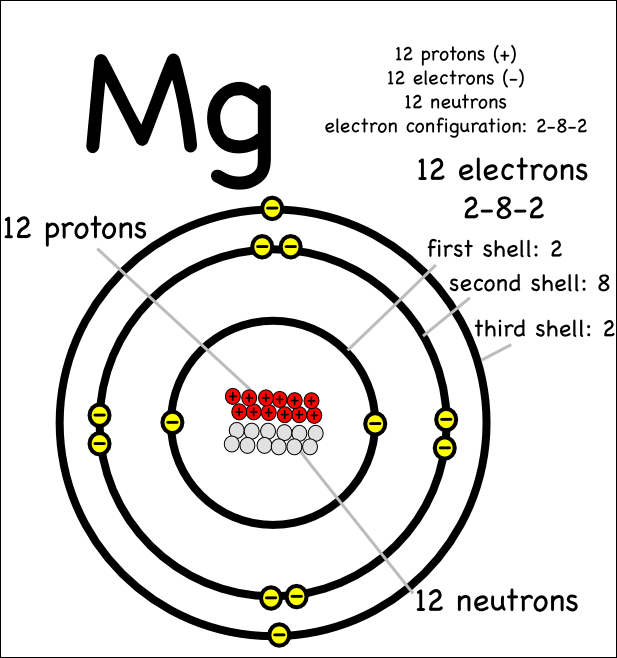
Magnesium Orbital Diagram What is the Electron Configuration of Lawrencium? Ground state magnesium orbital diagramġs 2 2s 2 2p 6 3s 2 is the ground state electronic configuration of Magnesium i.e., neither gain nor loss of electrons. 3s 13p 1 is the excited state electron configuration of Mg, where one electron is transferred from 3s to 3p to get an excited state. Excited state of magnesium electron configuration 3s 2 is the ground state configuration of Mg. Ground state magnesium electron configuration Magnesium unabbreviated electron configurationġs 2 2s 2 2p 6 3s 2 is an unabbreviated electronic configuration of magnesium. 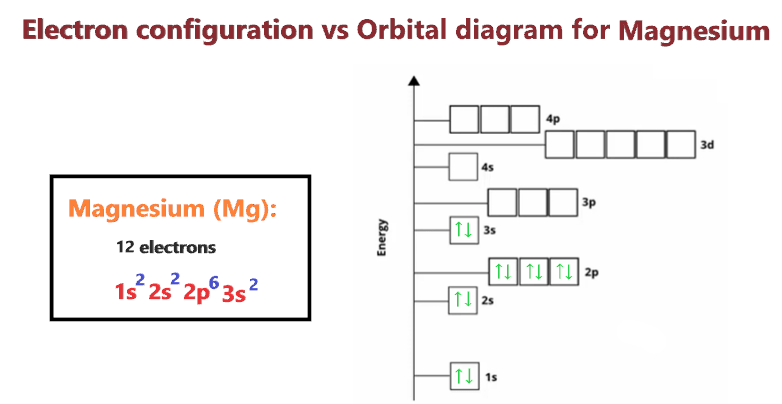
Magnesium Electronic Configuration Diagram Magnesium electron configuration notationģs 2 is the electronic configuration notation of Mg, where it has 12 electrons out of which 10 electrons can be represented for Neon and remaining two electrons in 3s. It is depicted in the form given below where filling of orbitals around nucleus takes place in increasing order of energy. The electronic configuration of Mg is 1s 2 2s 2 2p 6 3s 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
